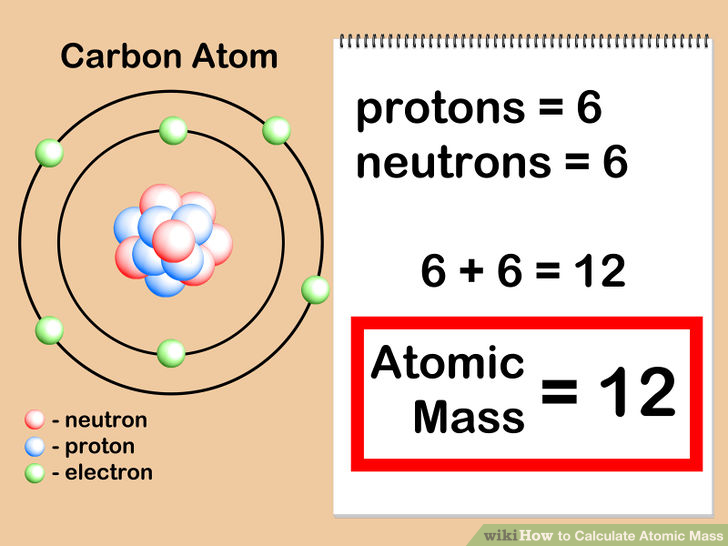
The mass of a proton is almost exactly the same as the mass of a neutron. nucleus of an atom of the carbon-13 isotope contains 13 nucleons.nucleus of an atom of the carbon-12 isotope contains 12 nucleons.So, the mass number (A) tells us the number of nucleons in the nucleus of an atom of the isotope of the element. Nucleon is the term used to describe both protons and neutrons. The mass number (A) of an isotope tells us how many protons and neutrons are in the nucleus of an atom of this isotope. We can estimate the mass of any isotope of an element, its isotopic mass, using its mass number (A). The relative atomic mass of an atom of carbon-13 is found to be 1.08333 times the mass of a carbon-12 atom, that is 1.083 × 12 = 13.00 The relative atomic mass of a carbon-12 atom is defined as 12.00 Play the game now! Estimating Isotopic Mass The table below gives the isotopic abundances for some elements on Earth: This means that if you take a lump of coal from nature, 98.90% of the carbon in the coal will be atoms of the carbon-12 isotope, while only 1.10% of it will be atoms of the carbon-13 isotope. The abundance of the carbon-12 isotope in naturally occurring bulk carbon is 98.90% while the abundance of the carbon-13 isotope in nature is 1.10% We call this amount of each isotope found in the naturally occurring element its abundance, or its isotopic abundance to be more precise. If you were to take a sample of carbon atoms, for example the soot from a chimney or a lump of coal, you would find that most of the carbon atoms are the carbon-12 isotope and only a few would be the carbon-13 isotope. 
The element carbon, for example, exists in nature as a mixture of different isotopes: stable 1 carbon-12 atoms and carbon-13 atoms. Most elements occur in nature as a mixture of different isotopes. No ads = no money for us = no free stuff for you! Abundance of Naturally Occurring Isotopes Note that we can measure the mass of each isotope and its abundance using Mass Spectroscopy.Then, let r.a.m = relative atomic mass of the element: Given the relative atomic mass (r.a.m.) of an element and the estimated mass of each of its isotopes, we can then estimate the relative abundance of each isotope:.We can estmate the the relative atomic mass (atomic weight) of an element E with the naturally occurring isotopes aE, bE, cE, etc, and with the respective abundances of A%, B%, C% etc,. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
